Thermal Inkjet Printers: Empowering Pharma Serialization and Compliance with Track and Trace Regulations
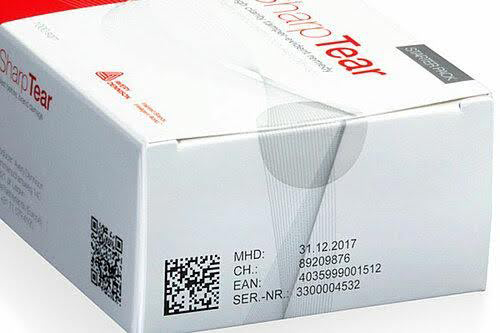
In the pharmaceutical industry, the implementation of serialization and track and trace regulations has become imperative to combat the rising threat of counterfeit medicines. Serialization, the process of assigning a unique and encrypted identifier to each product, facilitates traceability throughout the supply chain, safeguarding both the industry and consumers. The USA, in particular, is navigating these regulations through the Drug Supply Chain Security Act (DSCSA), aiming to establish a robust traceability system by 2023. In this landscape, our thermal inkjet printers, including Marokprint’s X1JET, Integra, and X2/X4 jet, as well as Reiner’s 1025, emerge as critical tools enabling pharmaceutical companies to meet regulatory requirements while saving time, money, and maintaining cleanliness in the manufacturing environment.
The Role of Serialization in Combating Counterfeiting
 Serialization acts as a powerful weapon against product counterfeiting, contributing significantly to industries like pharmaceuticals, cosmetics, food, and electronics. Each product in the production line receives a unique, encrypted number stored in a database, allowing for secure traceability. This not only aids in authenticating original products but also unveils counterfeit ones. With serialization data, the pharmaceutical industry gains the ability to identify genuine products, ensuring consumer safety.
Serialization acts as a powerful weapon against product counterfeiting, contributing significantly to industries like pharmaceuticals, cosmetics, food, and electronics. Each product in the production line receives a unique, encrypted number stored in a database, allowing for secure traceability. This not only aids in authenticating original products but also unveils counterfeit ones. With serialization data, the pharmaceutical industry gains the ability to identify genuine products, ensuring consumer safety.
Pharma Serialization in the USA
The pharmaceutical industry in the USA faces stringent serialization requirements outlined by the DSCSA. This regulation mandates drug manufacturers, repackagers, wholesale distributors, and dispensers to affix a product identifier to each package of prescription medication. AMI’s thermal inkjet printers play a pivotal role in meeting these requirements by providing efficient and reliable marking and coding solutions.
Choosing the Right Marking and Coding Solution
Selecting an appropriate marking and coding solution is crucial for successful serialization. Factors such as packaging material properties, abrasion resistance, contrast between marking and packaging surface, production speed, and IP protection class must be considered. AMI’s range of thermal inkjet printers ensures versatility, allowing pharmaceutical companies to choose the system and ink combination that best suits their specific requirements.


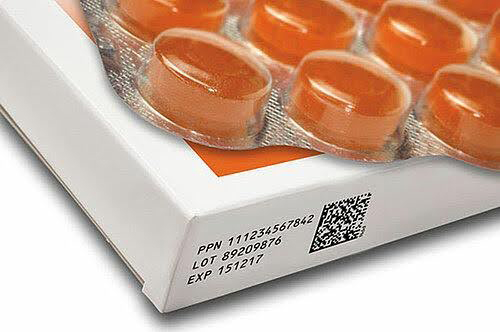
Benefits of Compliance
Complying with pharmaceutical serialization requirements offers numerous benefits beyond mere regulatory adherence. Serialization data becomes a valuable asset for supply chain visibility, demand forecasting, inventory management, order fulfillment, and product recall management. It enhances pharmaceutical quality control, streamlines procurement processes, and contributes to overall operational excellence.
Our Contribution
Our thermal inkjet printers are used extensively in the pharmaceutical industry and contribute significantly to achieving compliance with serialization regulations. These printers provide the necessary flexibility, precision, and speed required for efficient serialization in pharmaceutical manufacturing. Their reliability ensures that pharmaceutical companies can meet regulatory deadlines without compromising on quality.
As pharmaceutical serialization requirements continue to evolve globally, it is crucial for companies to invest in reliable and adaptable marking and coding solutions. AMI’s thermal inkjet printers stand out as essential tools, empowering pharmaceutical companies to navigate serialization and track and trace regulations seamlessly. By choosing us, companies not only ensure compliance but also enhance operational efficiency, protect consumers, and contribute to the overall integrity of the pharmaceutical supply chain.

